PROSILICO is a Swedish company focusing on the research and development of innovative technologies to provide high quality estimates of human ADME/PK (Absorption, Distribution, Metabolism, Excretion, PharmacoKinetics) directly from chemical structure.
PROSILICO’s unique data base of human clinical data provides the foundation to develop a wide scope of relevant prediction models for human ADME/PK. State of the art machine learning and AI is applied to extract the relevant information.
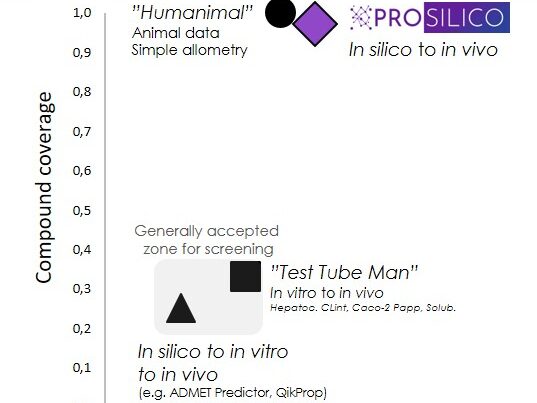
The PROSILICO platform has been validated by cross-validation, forward-looking predictions of new drugs on the market, and by blinded external validation.